Rosacea & Hidradenitis Suppurativa
Rosacea is a chronic inflammatory skin disorder that significantly impacts quality of life, yet its pathogenesis remains incompletely understood. Our laboratory has established a comprehensive research framework spanning metabolomics, single-cell transcriptomics, neurogenic signaling, and genetics to uncover the multi-dimensional mechanisms underlying this disease.
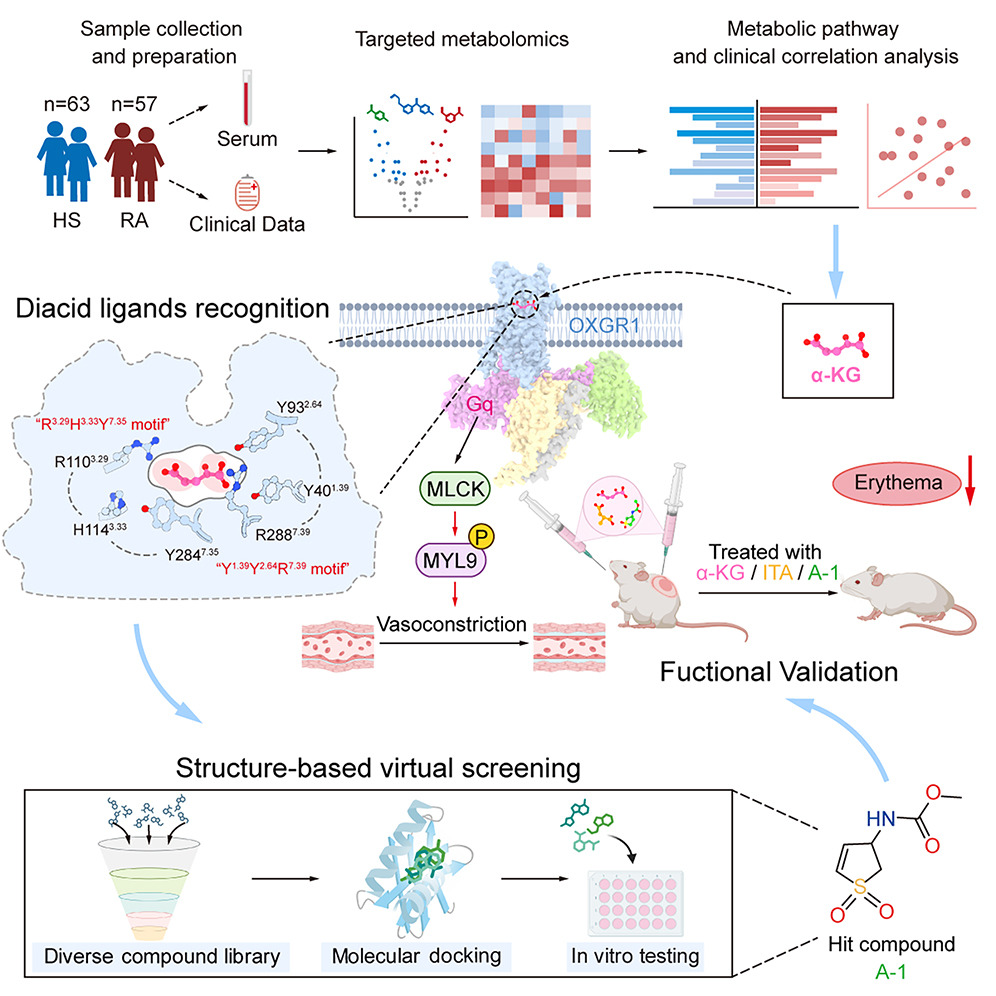
A landmark study identified alpha-ketoglutarate (α-KG) as a rosacea-associated metabolite that activates OXGR1, a GPCR enriched in vascular smooth muscle cells, promoting vasoconstriction and limiting pathological vasodilation. Cryo-electron microscopy structures of the OXGR1–Gq complex guided the development of A-1, a synthetic selective agonist that mitigates erythema with efficacy comparable to first-line therapy (Cell 2026). At the cellular level, single-cell transcriptomics revealed that pro-inflammatory fibroblasts are the leading source of vasodilative signals in rosacea lesions, with PTGDS depletion blocking disease development in mice (Nat Commun 2024). Complementing this, we demonstrated that high-sensitivity sensory neurons activate γδ T cells via a CGRP–RAMP1 axis, establishing a neuro-immune circuit that drives facial hypersensitivity (Nat Commun 2024).
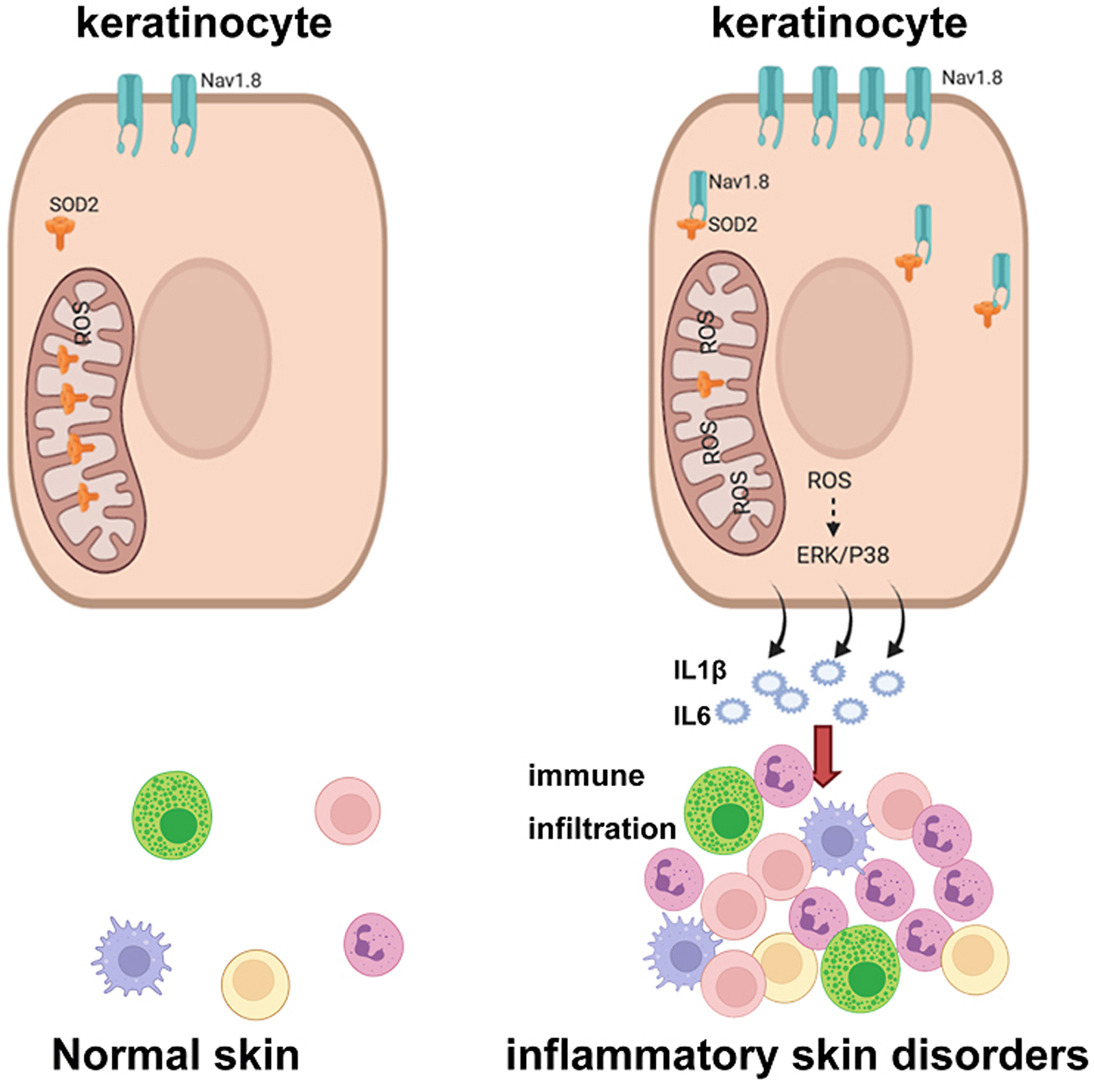
Genetically, whole-genome sequencing of rosacea families identified rare variants in LRRC4, SH3PXD2A, and SLC26A8 that converge on neural synaptic processes and promote excessive neuropeptide release (Nat Commun 2023). Serum proteomic profiling further resolved two distinct molecular endotypes—inflammatory-predominant and neurogenic-metabolic—providing a molecular foundation for personalized treatment (JAAD 2026). Key inflammatory pathways including STAT3/IL-36G (J Adv Res 2025), KRT6A/JAK1-STAT3 (J Biomed Sci 2025), and Nav1.8-mediated ROS signaling (Redox Biol 2022) have been dissected, while novel nanozyme-based ROS scavengers offer new therapeutic paradigms (Redox Biol 2024).
These mechanistic insights have been translated into clinical practice through multicenter randomized controlled trials, demonstrating that gabapentin improves rosacea flushing (JAAD 2025), paroxetine effectively treats refractory erythema (JAAD 2023), and hydroxychloroquine is a safe and efficacious treatment option for rosacea (JAAD 2021).
Hair Disorders & Regeneration
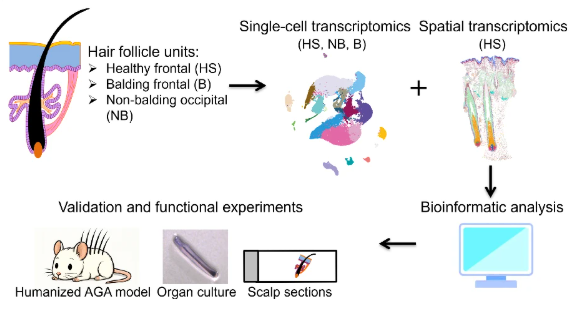
Androgenetic alopecia (AGA) manifests as progressive hair follicle miniaturization, yet the molecular mechanisms driving follicle miniaturization have remained elusive. Our single-cell transcriptomic atlas of human AGA anagen follicles revealed that hypercontractility of the connective tissue sheath (CTS) activates the mechanosensitive channel PIEZO1, inducing ectopic apoptosis of HF progenitor cells and driving progressive miniaturization. Pharmacological inhibition via the MLCK inhibitor ML-7 restored hair growth in patient-derived ex vivo models and humanized mice (Nat Commun 2026).
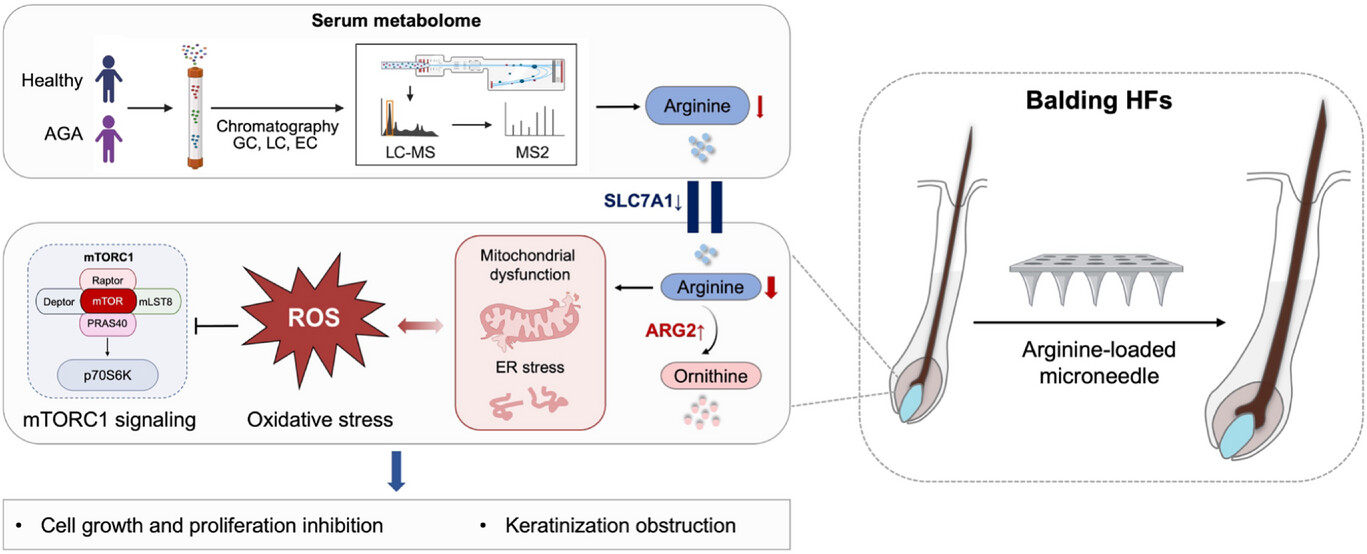
Metabolic profiling further identified arginine deficiency as a core pathogenic driver: downregulation of the transporter SLC7A1 and upregulation of ARG2 trigger ROS accumulation and mTOR inactivation, while microneedle-based arginine replenishment restores hair growth (Adv Sci 2025).
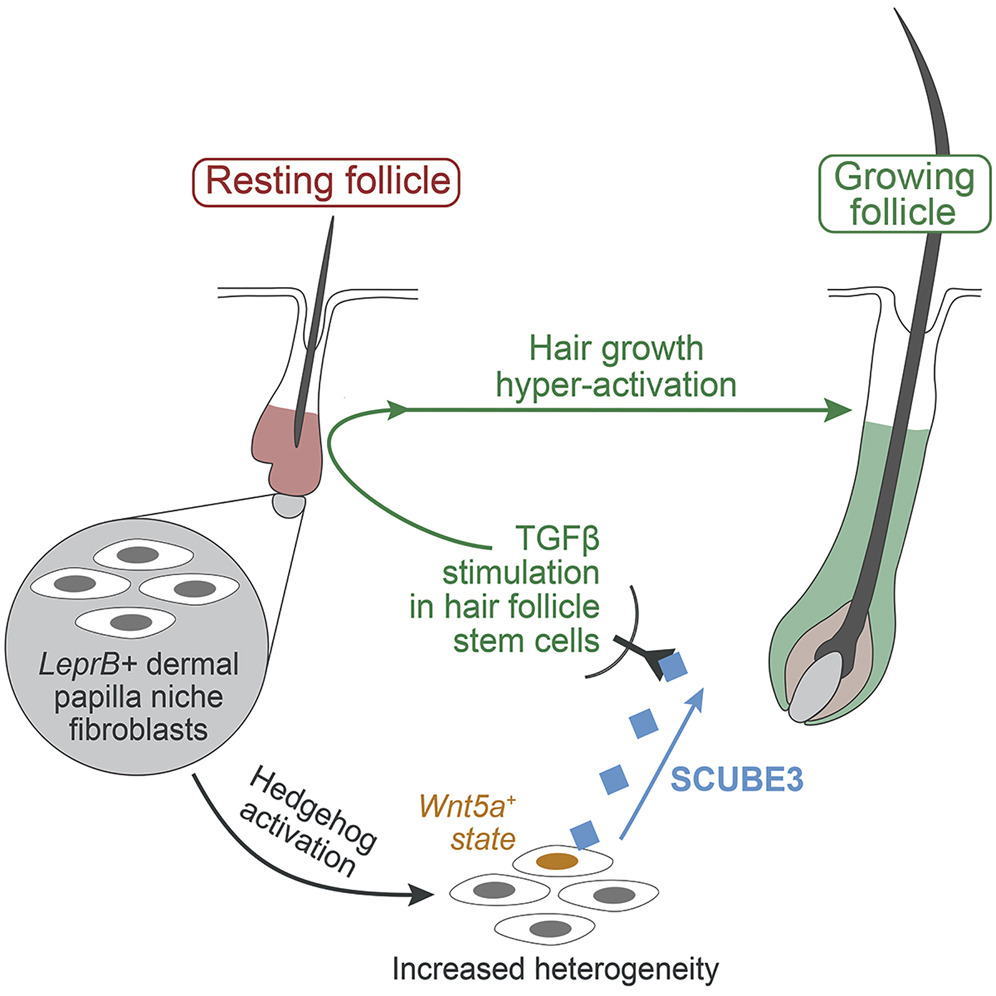
In hair pigmentation, skin organoids derived from melanocyte progenitors revealed a two-step patterning mechanism for establishing pigment patterns: COL6A3–CD44 signaling maintains organotypic melanocytes during culture, while SEMA3C–NRP1 signaling guides their migration to the bulge niche during post-transplant morphogenesis (Adv Sci 2025). In hair follicle regeneration, Hedgehog overactivation in the dermal papilla niche reprograms surrounding fibroblasts to produce SCUBE3, a TGF-β ligand sufficient to induce new hair growth (Dev Cell 2022).
For alopecia areata, a series of real-world clinical studies have established JAK inhibitor–based management strategies: ritlecitinib demonstrated robust efficacy across different anatomical sites, in adolescent populations, and in patients refractory to prior JAK inhibitor therapy, while a patient-centered tofacitinib dose-tapering protocol sustained treatment response without compromising efficacy (JAAD 2025; JAAD 2026; JAAD 2026; J Cutan Med Surg 2026).
Skin Aging
Aging is a complex, multifactorial process that affects every organ, with the skin serving as one of the most visible and accessible models for studying its biology. Our laboratory investigates the fundamental mechanisms of skin aging through an integrated approach combining longevity population cohorts, multi-omics technologies, and advanced cellular and animal models.
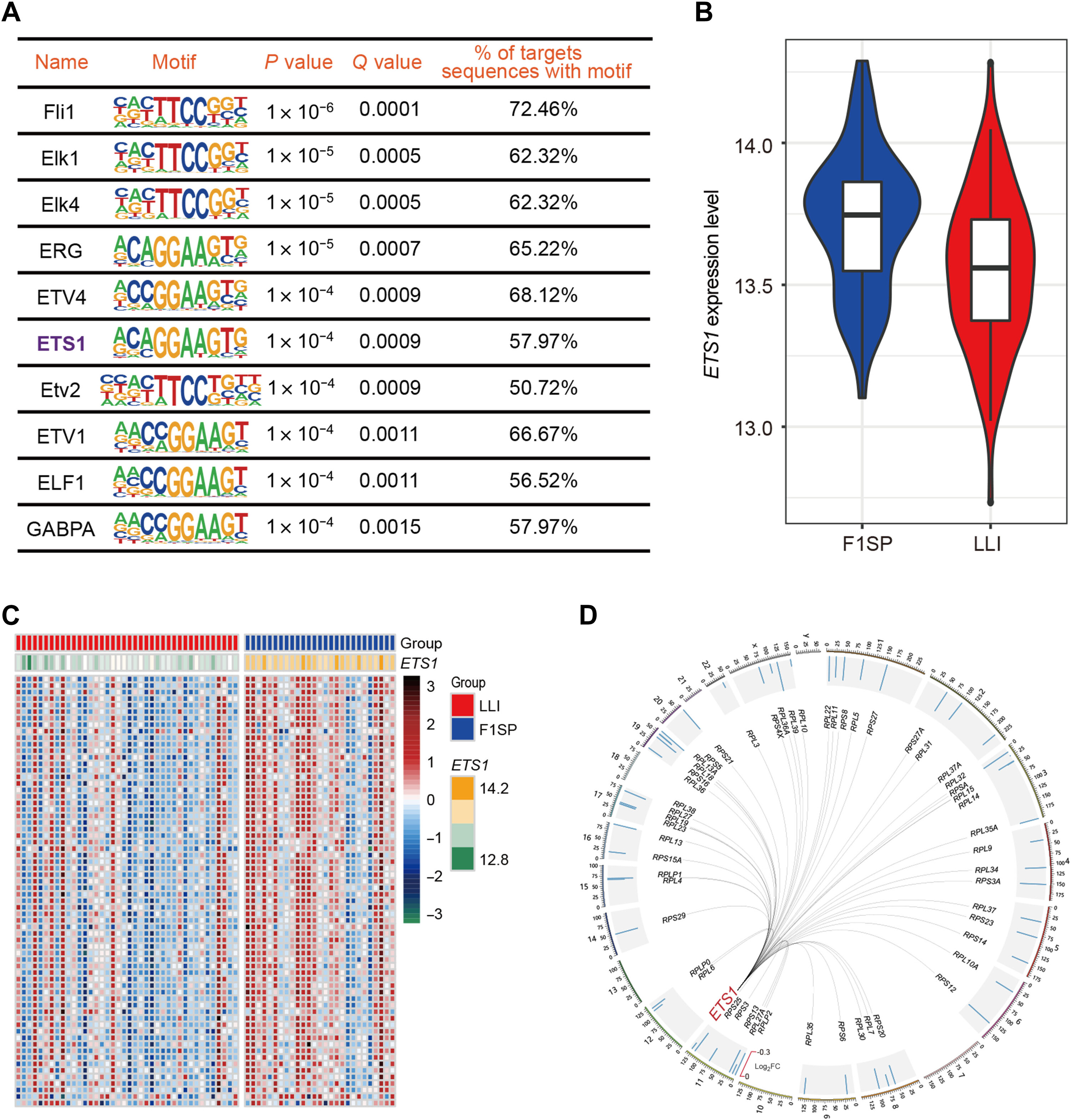
As a core member of the Aging Biomarker Consortium—a national initiative to establish standardized aging biomarkers across organ systems in China (Nat Med 2023)—we leverage large-scale cohort data to decode aging trajectories. A key discovery from longevity cohort transcriptomics revealed that ETS1 governs healthy aging by downregulating ribosomal protein gene expression, reducing energy-intensive ribosomal biogenesis as an adaptive energy-saving mechanism (Sci Adv 2022).
At the epigenetic level, we uncovered that depletion of the m6A demethylase FTO drives heterochromatin loss through a KAT8/RSF1 acetylation cascade, directly linking RNA modification to chromatin remodeling in skin aging (MedComm 2025). The transcription factor regulatory network has been further elucidated with the discovery that HOXC10 delays fibroblast senescence via the FZD6/Wnt/β-catenin pathway, with simvastatin identified as a functional mimic through Connectivity Map screening (Research 2025).
These foundational discoveries are driving the rational design of anti-aging therapeutics, with the overarching goal of translating molecular insights from longevity populations and cellular senescence mechanisms into effective clinical interventions.